|
This latter interaction means that the targeting of nascent polypeptides to the ER membrane is regulated by three GTPases that is, by SRP54 and both the α-subunit and β-subunit of the SRP receptor. The SRP54 subunit binds to ER signal sequences via its methionine-rich carboxy-terminal region (M-domain) and interacts with the SRP receptor complex via its central GTP binding domain. The SRP 9 kDa and 14 kDa proteins form a heterodimer that is involved in the SRP-mediated translation arrest or retardation, while the SRP 19 kDa protein facilitates the binding of the 54 kDa subunit of the signal recognition particle (SRP54) to the 7S RNA. Mammalian SRP consists of a 7S RNA and six proteins of molecular weight 9 kDa, 14 kDa, 19 kDa, 54 kDa, 68 kDa and 72 kDa. Translation of the targeted nascent chain resumes and the free SRP can enter a new cycle of targeting, while the signal sequence inserts into the ER translocon and is cleaved on the luminal side of the membrane by signal peptidase when a suitable site is available The binding of SRP to the SRP receptor allows the release of the signal sequence from SRP in a process that is dependent upon GTP binding and hydrolysis and that also requires the presence of the Sec61 translocon. This co-translational binding of SRP causes an arrest or retardation of translation that is relieved upon the interaction of the nascent chain/ribosome/SRP complex with the SRP receptor complex located in the ER membrane. These ER targeting signals are recognized and bound by a small ribonucleoprotein complex, the signal recognition particle (SRP), as soon as they emerge from the ribosome during protein synthesis. The signal sequence is often located towards the amino terminus of the protein, and in the case of presecretory proteins it is proteolytically removed after targeting is completed. This observation supports a model where the binding of the homologous GTPase domains of SRP54 and the α-subunit of the SRP receptor to each other regulates the release of ER signal sequences from the SRP54 M-domain.īoth secretory and membrane proteins destined for entry into the eukaryotic secretory pathway carry hydrophobic signal sequences that direct them to the endoplasmic reticulum (ER). When the effect of anti-SRP autoantibodies on protein targeting the ER membrane was further investigated, we found that the autoantibodies prevent the SRP receptor-mediated release of ER signal sequences from the SRP54 subunit. Nevertheless, anti-SRP54 autoantibodies interfere with signal-sequence binding to SRP54, most probably by steric hindrance. 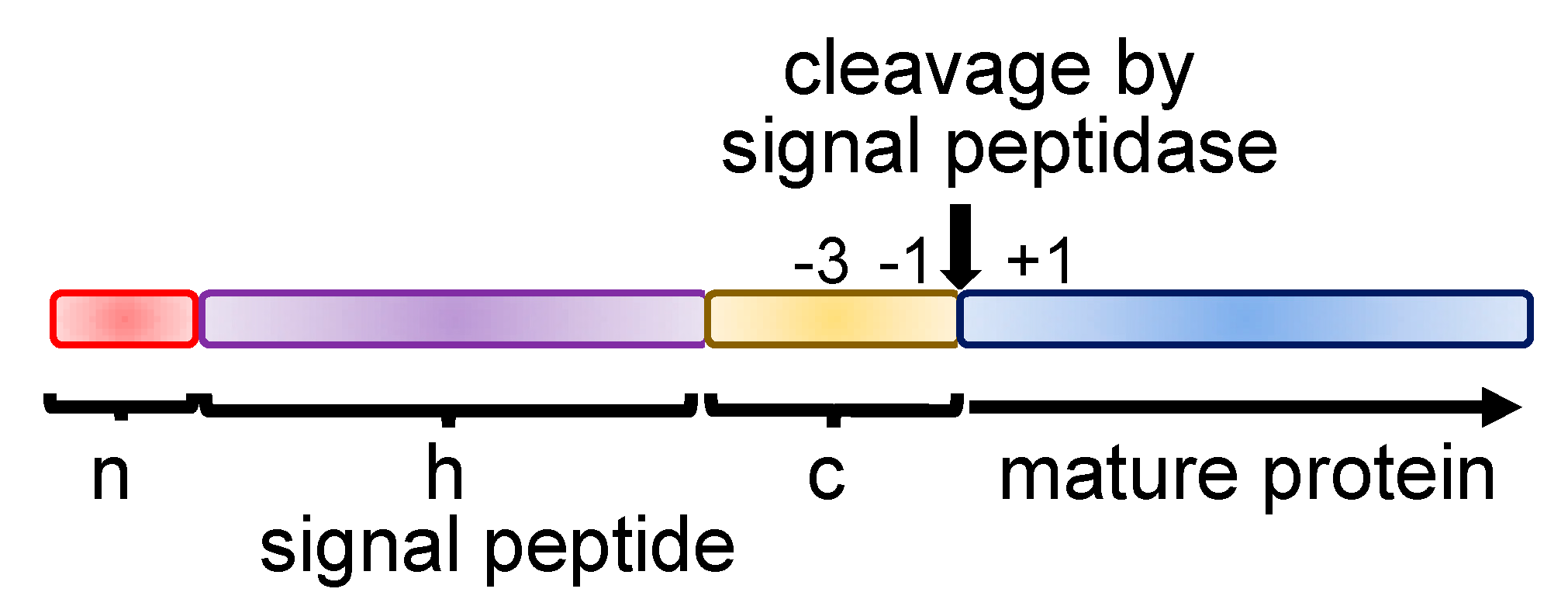
Our mapping studies showed that the anti-SRP54 autoantibodies bind to the amino-terminal SRP54 N-domain and to the central SRP54 G-domain, but do not bind to the carboxy-terminal M-domain that is known to bind ER signal sequences. We found that the anti-SRP54 autoantibodies had a pronounced and specific inhibitory effect upon the translocation of the secretory protein preprolactin when analysed using a cell-free system. 
In the current article, we have characterized the specificity of anti-SRP54 autoantibodies, which are highly characteristic of polymyositis patients, and investigated the effect of these autoantibodies on the SRP function in vitro. At the ER membrane, the binding of the signal recognition particle (SRP) to its receptor triggers the release of SRP54 from its bound signal sequence and the nascent polypeptide is transferred to the Sec61 translocon for insertion into, or translocation across, the ER membrane. The 54 kDa subunit of the signal recognition particle (SRP54) binds to the signal sequences of nascent secretory and membrane proteins and it contributes to the targeting of these precursors to the membrane of the endoplasmic reticulum (ER).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
